Potions and Poisons
| Potions and Poisons | |||||||
|---|---|---|---|---|---|---|---|
| Type | Chemistry | ||||||
| Category | Lab | ||||||
| Description | This event is about chemical properties and effects of specified toxic and therapeutic chemical substances, with a focus on household and environmental toxins or poisons. | ||||||
| Event Information | |||||||
| Participants | 2 | ||||||
| Eye Protection | Category C | ||||||
| Allowed Resources |
| ||||||
| Approx. Time | 50 minutes | ||||||
| History | |||||||
| First Appearance | 2018 | ||||||
| Latest Appearance | 2026 | ||||||
| Rotates | Yes | ||||||
| Forum Threads | |||||||
| |||||||
| Question Marathon Threads | |||||||
| |||||||
| Official Resources | |||||||
| Division B Website | soinc | ||||||
| Division B Results | |||||||
| |||||||
Potions and Poisons is a Division B event for the 2026 season. In Potions and Poisons, participants demonstrate their knowledge on specified substances, and chemical properties and effects with a focus on common toxins and poisons.
General Information

The event consists of both a written exam and hands on lab activities, which may be performed by students or run as a demonstration. Exam questions contribute 60% to the final score, while lab exercises and questions are worth 40%.
Category C goggles are required as well as a lab coat or apron. Skin must be covered with long sleeves, long pants, and closed-toe shoes. Hair longer than shoulder-length must be tied back.
Each student may bring one note sheet and a class II calculator. Teams are encouraged to bring a set of specific recommended lab equipment. Teams that do not bring this equipment may not be able to complete some or all of the lab activities, though they may still complete the written exam portion.
Topics covered
Topics for the written exam include:
- Ionic/Covalent bonds with relation to conductivity
- Mixtures, solutions and compounds and separation of the components within them
- Chemical and physical properties and changes
- Balancing chemical equations
- Dilution's effect on toxicity
- Toxic spills and effects of their spread via water, wind, or gravity
- Identification of various poisonous organisms along with their toxic effects (listed on the rules)
- Effects/chemistry of common household toxins and chemicals
Lab Tasks
Activities for the lab portion of the event include:
- Chromatography
- Mixtures of reagents
- Separation of mixtures
- Serial dilutions
- Measuring pH
- Conductivity testing
Lab Equipment
For current information, always check soinc.org.
Competitors may bring a kit of lab equipment as specified on the Recommended Lab Equipment list for the current season. The 2025 list may be found near the end of the rules manual or on the soinc.org event page. While not all of the items listed may be needed during the event, it is better to err on the side of caution and be prepared for anything. If equipment is required but a competitor did not bring it to the event, it is unlikely that an event supervisor will be able to provide it. Notable equipment for this event includes Erlenmeyer flasks, graduated cylinders, test tubes, beakers, spot plates, petri dishes, and testing materials such as pH paper and conductivity meters. Some tools for handling chemicals may also be required, such as tweezers, spatulas, and stir rods.
Lab Tasks
Tasks that students may be asked to perform and may be asked questions about include:
Chromatography
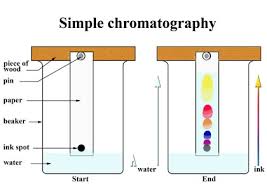
Chromatography is when a mixture (i.e. markers, pens, organic juices) is separated by passing it through a solution, which will most likely be water. There are many different types of chromatography such as gas or column chromatography, but in the lab task, simple chromatography is the most important.
To start, draw a start line and the ink spot on the chromatography paper. The start line should be above or near the water line, ~2-3 cm from the bottom of the paper for accurate results. Then, attach the paper to a stick or lean it against a stick in the beaker. The solution (water) should travel up the paper and separate the sample into different colors. After roughly 10-20 minutes depending on the paper, the chromatography should be done and the paper should be taken out to dry. After that, mark the water line's endpoint and where the colors ended. It is important to distinguish the number of colors that are there and use the retention factor for each color. To find the distance, put dots at the highest point of each color and measure it.
The retention/retardation factor (RF) is the distance moved by the compound divided by the distance moved by the solute. In other words, divide the distance between the color and the start over the distance the water traveled.
Mixtures of Reagents
Reagents are basically the starting materials used in chemical reactions. They're used to test if a reaction would occur. This type of lab is rare, but it can occur here and there.
These types of labs depend solely on the situation and doesn't have a set procedure like the other labs. On the test, it should describe the exacts about the lab and what to do.
Separation of Mixtures
This type of lab, like the previous example, depends solely on the situation. These mixtures are reversible, and the most important thing is determining the proper separation method to use.
Here is a list of different separation methods:
- Filtration - separating an insoluble solid from a liquid using a filter
- Chromatography - identify chemicals (coloring agents) in foods or inks through polarity
- Evaporation - separating a solution of a solute and a solvent by evaporating the liquid
- Simple Distillation - separating a liquid from a solution and saving it
- Fractional distillation - separating a solution of two miscible liquids
- Magnetism - separating mixtures with one part having magnetic properties
- Separating funnel - immiscible(i.e. not forming a homogenous mixture when mixed) liquids can be separated using their density. This process uses a funnel-like device.
- Centrifuging - fast spinning machine that can separate solids from liquids
- Crystallization - separating solids by making them crystallize
- Sedimentation - separating solids from liquid by letting it settle
- Precipitation - creating a solid from a solution
- Sieving - using particle size to separate mixtures by using a sieve
- Decantation - separating liquids or homogenous mixture by letting one part settle and pouring the liquid out.
- Leaching - extracting a solid by dissolving it in liquid
- Winnowing - separating lighter solids from heavier ones use wind.
Dil
☃☃whereC1 = the initial concentration of the soluteV1 = the initial volume of the solutionC2 = the final concentration of the soluteV2 = the final volume of the solutionFor example, if a solution has a 10% concentration of salt in one liter of water, adding another liter of water would halve the concentration of the salt, to 5%.↵This example can be shown mathematically using the above equation, where:C1 = 10%V1 = 1 LC2 = 5%V2 = 2 L☃☃☃☃☃☃For example: when told to make a sample with a 1:1440 dilution factor, take the solution and dilute it by 12, 12, and 10 respectively (12 * 12 * 10 = 1440) to get to that certain dilution.pH determinationpH is the potential of hydrogen, which basically is a number that decides how strong of a base or acid a solution is. The range of pH in water is 0-14, however, with other substances, it can go well below 0 or well above 14. In this event, the 1-14 scale will likely be used. 0-6 pH is acidic, 7 is neutral, and 8-14 are bases.To test the pH, litmus or pH paper are typically used. There are many ways of doing this, but the one that's most efficient is just dipping the paper in the water (about 10% of the paper should be submerged) and taking it out to dry on a spot plate. Then, match the color of the paper to the color on the litmus paper box/holder. If the paper just looks wet, it is neutral. Litmus/pH paper does not work with heavily dyed or dark colored liquids.In pH, every number down (relative to water's scale) is 10 times stronger than the previous number. (I.e pH 3 is 10x stronger than pH 4; pH 4 is 100x stronger than pH 6; pH 6 is .01x stronger than pH 4)ConductivityConductivity tests are performed with a conductivity meter, which can either be bought or made. Cheaper conductivity meters typically use LEDs to indicate the conductivity of a liquid, where a dimmer LED means the liquid is less conductive and a brighter LED means that it is more conductive. While these types of meters are cheaper, they typically require more work as they do not display a number for the conductivity. Electronic conductivity meters are typically more expensive, but display a number as opposed to just turning on a LED. Not all conductivity meters are the same, and it is important to read the manual that comes with it to know how it works. Avoid getting liquids anywhere but the probes, and be sure to wipe off the meter after use.Ionic bonds are conductive, while covalent bonds are not. If a compound has a lot of ionic bonds, it will be more highly conductive than a substance that does not.The AtomAtoms are the particles that make up elements. This means that atoms come together to make everything on Earth, regardless of what it is or where it came from. The particles that make up these atoms (called subatomic particles) determine their properties, such as how reactive they are.Subatomic ParticlesAtoms are made of three particles: protons, electrons, and neutrons.Protons are found in the center of the atom, known as the nucleus. The number of protons in the nucleus of an atom is the same as its atomic number, and determines what element it is. No two elements will have the same amount of protons, and as a result will not have the same atomic number. Protons are also positively charged and weigh the most out of these three particles.Neutrons are also found in the center of the atom, though they have no electric charge. Their mass is slightly larger than that of a proton, though the mass of each particle is commonly referred to as an atomic mass unit. The atomic mass of an element is found by adding together the amount of protons and neutrons in the nucleus. While the amount of protons determines the identity of an element, the number of protons may change. These variations of an element are known as isotopes, and are identified by their atomic mass. For example - carbon-12 has six protons and six neutrons in its nucleus which gives it an atomic mass of 12. This isotope makes up a majority of elemental carbon found on Earth, but other isotopes such as carbon-13 and carbon-14 also occur naturally.Electrons are the smallest of these three particles, orbiting around the nucleus instead of being a part of it. Electrons are negatively charged, and play a major role in how elements interact.Shells, Subshells, and OrbitalsA Bohr model of a neon atom with labeled subshells.There are a variety of different models that explain how an atom is structured. The Rutherford-Bohr (or simply Bohr) model of the atom is one of the simplest and most common - in it, the atom is depicted as a central nucleus with electrons in layers known as shells at different distances from the nucleus. These shells have different energy levels, with the one closest to the nucleus having the lowest energy level. The first shell only holds two electrons, the second shell holds eight, the third shell holds 18, and so on. A general rule of thumb is that each successive shell can hold ☃☃ electrons where n is the shell's number. The outermost shell of an atom is known as the valence shell, and its electrons are known as valence electrons. In the Bohr model of a neon atom pictured to the right, all eight electrons in the second shell are valence electrons. Atoms tend to gravitate towards being very stable, having as many full shells as possible. If an atom is missing one electron in its outermost (valence) shell, it will be easier for it to bond to an element with one valence electron. Conversely, if an atom only has one valence electron it will be more willing to "give it away" or bond with an atom that needs one more electron to fill its valence shell. While for many elements the outermost shell can hold more than eight electrons, oftentimes elements bond in a way that results in each atom having eight electrons in its outermost shell. This is known as the octet rule.While the Bohr model is useful in explaining the energy levels and reactivity of certain elements, it leaves a lot to be desired when explaining how electrons move around the nucleus. Many electrons do not circle the nucleus at all, instead filling up spaces known as orbitals. Electrons are very small and move very quickly, and as a result it is difficult to determine where an electron is at a given point in time. However, these orbitals are where an electron spends a majority of its time. While the Bohr model simplifies the placement of electrons in space, it can be expanded upon in order to paint a better picture of how electrons move and interact with other particles. Electron shells as described above can be broken down into subshells, which are just sets of one or more orbitals. These subshells are known as s, p, d, and f. Subshells are filled in that order, starting with s and moving up through p. The s subshell holds 2 electrons, the p subshell holds 6, the d subshell holds 10, and the f subshell holds 14 electrons.Electron ConfigurationThe electron configuration of an atom has to do with subshells and orbitals, listing out where the electrons of an atom are likely to be located. For example, the electron configuration of neon is ☃☃. The large number is the shell the electron is located in, the letter is the subshell it is located in, and the superscript describes how many electrons are in the subshell.Looking again at the Bohr model of neon, it has two electrons in the first shell and eight in the second. Because electrons fill the smaller subshells first, this means that the first shell can be written as ☃☃. Each shell is filled in the same way. Because the s subshell can hold two electrons and the p subshell can hold six, the second shell can be written as ☃☃. Putting the two shells together gives the complete electron configuration of neon which is ☃☃.The Periodic TableFor additional information about the periodic table, please see Chemistry Lab/Periodicity.Intramolecular bondingA chemical bond is an attraction between two atoms that causes them to bond together, which can create molecules. The two types of bonding that the current version of the Potions and Poisons rules says to know are ionic and covalent bonding. Bonding occurs with electrons, in which the electrons are taken (ionic) or shared between atoms (covalent).ElectronegativityAn electronegativity tableElectronegativity is a property of elements that is defined as the tendency of an atom to attract electrons. The difference between two atoms' electronegativities decides the type of bond they make. A chart known as an electronegativity chart can be used to find the electronegativities of different elements. Electronegativity increases from left to right and from bottom to top, therefore helium has the highest while francium has the lowest.Ionic BondingIonic bonding is a type of bonding in which one atom takes an electron from another atom. This type of bonding occurs when the difference in electronegativity is high (typically a difference of 1.7-2.0). A common example of this is the compound NaCl, or table salt. The metal Na (sodium) bonds with the halogen gas Cl (chlorine). When solid, ionic bonds are typically not conductive. However, when liquid, it is conductive.Ionic bonds are typically formed with a crystal lattice structure and have a high melting temperature. They're also water soluble.Shown is a Venn diagram which demonstrates the difference between Ionic and Covalent bondsTo name a simple ionic compound, use the name of the regular metal, followed by the name of the nonmetal, with the latter using the ending "-ide". For example, NaCl would be written as sodium chloride.Covalent BondingCovalent bonding is a type of bonding in which atoms 'share' electrons. This occurs when there is a low difference in electronegativity. Two very common examples are the molecules H2 and O2. Because the bonds are formed between two atoms of the same element, the difference in their electronegativities must be zero. Covalent bonds are liquids and gases, at room temperature (294֯ K), however, they are not conductive.Covalent bonds are formed with a true molecule structure and has a low melting temperature. They are not water soluble (most of the time) and are odorous.Covalent bonds can be nonpolar or polar. In polar covalent bonds, the atoms have different electronegativities, and therefore share electrons unequally. The more electronegative atom has a partial negative charge, while the less electronegative atom has a partial positive charge. In nonpolar covalent bonds, the atoms have similar electronegativities and therefore share the electrons equally. These are denoted with the δ symbol (Greek lowercase delta), using δ- for partial negative charge and δ+ for partial positive charge.To summarize, polar covalent bonds share electrons unequally and therefore have poles. Nonpolar covalent bonds share electrons equally, and therefore do not have poles.Intermolecular Forces and DipolesIntermolecular forces (IMF) are interactions between atoms or molecules that do not result from electronic bonds. They are comparatively very weak, but still play an important part in all forms of chemistry. The three main forms are London dispersion forces, dipole-dipole forces, and hydrogen bonds. All of these intermolecular forces can be referred to as van der Waals forces.When discussing intermolecular forces, the word dipole appears a lot. A dipole movement is a measurement of the separation of charges on a molecule. This quantity is written as a vector, since it has both direction and magnitude. Permanent dipoles occur due to covalent bonds as a result of electronegativity. These dipole movements are what separate polar and nonpolar molecules. Polar molecules will have positive and negative areas referred to as ☃☃ and ☃☃. Instantaneous dipoles occur in all molecules due to the movement of electrons. Since electrons are constantly moving, one part of an electron will always be more negatively charged. These instantaneous dipoles can attract each other, resulting in weak intermolecular forces.London DispersionLondon dispersion forces can also be referred to as instantaneous dipole–induced dipole forces, and are exhibited by all molecules. These forces come from interactions between uncharged atoms and molecules, and are the weakest types of intermolecular forces.Dipole-DipoleAlso known as Keesom forces, dipole-dipole interactions only occur in polar molecules. These forces occur when the ☃☃ area of a polar molecule is attracted to the ☃☃ area of another polar molecule.Hydrogen BondingThe strongest of the three types of IMFs, hydrogen bonds occur when a hydrogen atom is attracted to a strongly electronegative atom such as nitrogen, fluorine, or oxygen.Mixtures, Solutions, and Compounds/MoleculesMixturesMixtures are substances that are made by combining 2 or more mixtures. There are two types, homogenous and heterogenous. Homogenous mixtures are substances that have a uniform mixture throughout the substance. Some examples are lemonade, salt water, and air. Heterogenous mixtures are substances that have inconsistent component ratios throughout the whole. Basically, it's just not mixed very well. Some examples are trail mix, salad, and sand and pebbles.SolutionsSolutions are a type of mixture that requires a solute, a solid, and a solvent, a liquid. This type of mixture is often referred to as a homogenous mixture since the mixture is consistent throughout.Compounds/MoleculesCompounds are molecules that have two or more different elements that are chemically conjoined together. H2O, CO2, and H2O2 are all types of compounds. O2 and He2 are not. Molecules are just a group of elements that are chemically conjoined.Types of ChangesPhysical ChangesPhysical changes affect the appearance of a substance, not the chemical composition. Physical changes could be used to separate components of a mixture into their own individual parts. Examples of physical changes include tearing paper, boiling water, mixing sand and water, and melting an ice cube.Chemical ChangesChemical changes are the altering of a substance to form a new substance with a different chemical equation. Examples include burning a candle, digesting food, and rusting iron.Types of ReactionsSynthesis and CombustionA synthesis reaction is one of the simplest chemical reactions, occurring between either pure elements or compounds. Synthesis can also be referred to as combination, so pay attention to the wording on test questions. An example of synthesis is the formation of sodium chloride (table salt) using sodium metal and chlorine gas:☃☃However, not all synthesis reactions are this simple. While a majority of the time synthesis equations will only have one product, that is not always the case. Take the following equation for photosynthesis. The carbon dioxide and water are forming the more complex glucose, while the oxygen is left over.☃☃One specific type of synthesis is also known as combustion, and it takes place when a hydrocarbon reacts with oxygen. The product will always be carbon dioxide and water. For example:☃☃DecompositionJust as two components can be combined to form a single product, one product can be broken down to form its individual components. This is referred to as decomposition. It will always have one reactant and multiple products. In this example, hydrogen peroxide breaks down to form water and oxygen.☃☃Single DisplacementSingle displacement (also known as single replacement) is a type of reaction that occurs between one pure element and one compound. Generically, it can be written as ☃☃ For example, when putting magnesium into hydrochloric acid the following reaction will occur:☃☃Typically, single displacement occurs when A is more reactive than B, the element it is replacing. However, not all elements can replace each other. In order for a single displacement reaction to work, A and B have to either be different metals or halogens. The one exception to this rule as hydrogen, as seen in the equation above. If A and B are different metals then C must be a cation, or an ion with a positive charge. If they are halogens then C must be an anion, or an ion with a negative charge.Elements will always replace each other in a specific order, known as a reactivity or activity series. An element on the list can replace all elements below it, but not ones above.☃☃Double DisplacementDouble displacement (also known as a metathesis reaction or double replacement) is very similar to single displacement, but it occurs between two compounds instead of between one compound and one pure element. Typically these reactions occur in a solution, and either form an insoluble solid known as a precipitate or water. Take the following equation with potassium chloride and silver nitrate:☃☃Both of the reactants are aqueous, as well as one of the products (potassium nitrate). However, the silver chloride is a solid. Since the solid formed here is called a precipitate, this type of reaction is known as a precipitation reaction.Another type of double displacement reaction is known as a neutralization or acid-base reaction. Water is typically a product of these reactions, since the goal of a neutralization is to form products that are not acidic or basic.☃☃In the previous equation, the acid (☃☃) reacts with the base (☃☃) to form water and sodium sulfate.Chemical EquationsChemical reactions are written out as chemical equations. A chemical equation has two parts: reactants and products. The atoms in the reactants are rearranged to form the products.Chemical equations are written left-to-right with the products following the reactants, and an arrow sign (which is read as "yields") pointing from the reactants to the products. Each individual reactant/molecule is represented with a plus sign (+).↵The following equation is an example of a chemical equation, with the reactants on the left and the product on the right.☃☃If there are multiple instances of a molecule, the number of molecules is written as a coefficient. For example, the product in the above equation is water or H₂O. There are two water molecules present, which is written as 2H₂O.☃☃Balancing Chemical EquationsIn a chemical reaction, the quantity of each element cannot change (If there are n atoms of element A in the reactants, there must be n atoms in the product). A chemical equation must have equal quantities of each element on either side of the arrow. As mentioned above, adding coefficients to molecules can show that there are those many molecules present. However, if an equation is given without coefficients, chances are that there is inequality on either side.↵Consider the example given above; however this time it is without coefficients:☃☃Counting the number of each element on either side of the equation gives the following amounts:Hydrogen on left: 2Oxygen on left: 2Hydrogen on right: 2Oxygen on right: 1This cannot be a balanced equation, because the number of atoms is unequal. To fix this issue, it is necessary to balance the chemical equation.To balance a chemical equation, add coefficients to make the number of atoms of each element equal. For example, take again the previous equation:☃☃Notice that there is only one type of molecule as the product, meaning that it is the only molecule that a coefficient can be added to. A basic way to find the proper coefficient is to find a ratio between the two elements on one side and apply that to the other side. In this example, there are two times as many hydrogens as oxygens. In addition, all coefficients must be whole numbers. Therefore, the lowest coefficients would be a two in front of the hydrogen gas (on the left) and a two in front of the water.↵This gives us a balanced equation of:☃☃An easy way to go about this is to guess and check the distinct elements first. For example:☃☃ is to find a ratio between the two elements on one side and apply that to the other side. In this example, there are two times as many hydrogens as oxygens. In addition, all coefficients must be whole numbers. Therefore, the lowest coefficients would be a two in front of the hydrogen gas (on the left) and a two in front of the water. This gives us a balanced equation of:
An easy way to go about this is to guess and check the distinct elements first. For example:
Start with C, as it's only present in two compounds that don't involve O (which is involved in 3). If putting 3 at CO2 doesn't work, double it.
When the answer to the blank is 1, it should be left blank. Only fill in an answer if it is greater than one. When the equation is already balanced, just write "balanced".
Poisonous Plants and Animals
This section is incomplete. |
The poisonous plants and animals listed in the 2026 season rules are:
- Poison ivy (Toxicodendron radicans)
- giant hogweed (Heracleum mantegazzianum)
- Calabar bean (Physostigma venenosum)
- rhubarb (Rheum rhaponticum)
- American false hellebore (Veratrum viride)
- cane toad (Rhinella marina)
- poison dart frog (Dendrobatidae)
- Portuguese man o’ war (Physalia physalis)
- lionfish (Pterois sp)
- fat tail scorpion (Androctonus australis)
Poison Ivy (Toxicodendron radicans)

Poison ivy grows most regions of the US, typically in woods, fields, and along roadsides, especially where vegetation is disturbed. It can be identified by its three thin, pointy, and shiny leaves. The leaf color depends on the season. In the spring, the leaves are reddish; in the summer, green; in the fall, orange to bronze.
Upon contact with the oil from poison ivy, an allergic reaction happens. Touching the plant itself is not the only way to contact the oil; touching gardening equipment or pets that have contacted the ivy can also spread the oil. Symptoms of a reaction include itching, redness, swelling, and blisters. It is important to note that the blisters are NOT contagious.
The poison, Urushiol, is an oily mixture of organic compounds with allergenic properties. It can also be found in mango trees, lacquer trees, poison oak, and other Anacardiaceae plants. It gets its name from the Japanese word for the lacquer tree, urushi. The urushiol allows the tree to form a hard lacquer which is used in Asian lacquerware. It's a pale yellow liquid which has a boiling point of 200C. It can easily be removed with soap and water, provided that the skin hasn't absorbed it yet.
Giant Hogweed (Heracleum mantegazzianum)
Giant hogweed is a high weed that grows tall in zones of North America, Europe, and Asia, most often along roadsides, riverbanks, and fields. It grows to 14 feet tall with large, toothed leaves and an umbrella-like cluster of white flowers. Its purple-spotted, hollow stems are covered with small bristles.
Giant hogweed sap contact, with its furanocoumarins content, induces painful dermatitis-type reactions following sun exposure, referred to as phytophotodermatitis. The reaction can vary from painful blisters and erythema to scarring which could last for several years. Sap can penetrate through clothing or equipment and the minimal amount will have effects. If the skin has not absorbed it, immediate soap and water washing will reduce effects.
Calabar Bean (Physostigma venenosum)
The Calabar bean is a vine that twines, native to West Africa and inhabiting tropical forests and riverine environments. The seeds, which are dark brown or reddish-brown beans, are contained in pods. The plant is characterized by large, shiny leaves and purple-colored flowers.
Seeds contain physostigmine, a toxic cholinesterase-inhibiting alkaloid that disturbs nerve function. Ingestion produces nausea, muscular weakness, paralysis, and, in severe cases, death due to respiratory failure. The bean, in ordeal tests for guilt, was employed by traditional West Africans because the survivors were considered innocent. Treatment of exposure is prompt medical attention, and it has to be kept away from accidental ingestion.
Rhubarb (Rheum rhaponticum)
Rhubarb is a temperate perennial shrub cultivated in farms or gardens. Rhubarb produces large, triangular leaves and thick reddish stalks. Rhubarb stalks are edible and used in cooking, while leaves are toxic.
Rhubarb leaves hold anthraquinone glycosides and oxalic acid, which are toxic when ingested. Their effect is nausea, vomiting, and stomach pain, and, with large doses, renal toxicity or seizures. Toxins are not destroyed by cooking, and even small doses are hazardous. Avoid eating or handling the leaves, especially around children or pets.
American False Hellebore (Veratrum viride)
American false hellebore is found in meadows and wetlands in North America, particularly in mountain country. It has large, pleated green leaves and a tall spike with a congested cluster of star-shaped, greenish-yellow flowers.
The plant contains steroidal alkaloids such as veratridine, which are poisonous on ingestion. They are neurotoxicants and exhibit manifestations in the form of nausea, vomiting, hypotension, bradycardia, and in the most severe cases, cardiac arrest. Traditionally, it was used in low dosages for clinical requirements, but the drug is fatal if ingested accidentally when used mistakenly as food crops. Its contact with the skin with immediate washing of the skin and abstention from ingestion are critical.
Cane Toad (Rhinella marina)
The cane toad, native to Central and South America but now introduced to places like Australia, inhabits tropical and subtropical environments, typically near water. It has dry, warty skin that is typically brown or gray with large parotid glands behind the eyes.
These glands secrete bufotoxins, a mixture of cardioactive steroids, which would lead to extreme poisoning if touched or consumed. The symptoms and signs are drooling, convulsions, irregular heartbeat, and death in extreme cases, primarily in pets or animals that attempt to ingest the toad. The poison is transferred by skin or mucous membrane contact. Immediate washing off of areas that come into contact with water and medical or veterinary support should be obtained.
Poison Dart Frog (Dendrobatidae)
Poison dart frogs are small, vividly colored frogs from Central and South America that inhabit rainforests, typically displaying bright red, blue, or yellow colors with black spots. Their bright coloration is an indication of their toxicity.
Their skin secretes batrachotoxins, which are strong neurotoxins that disrupt muscle and nerve function. If contact or ingestion occurs, then symptoms will include paralysis of muscles, heart failure, and death. Indigenous people used these toxins to poison the tips of hunting darts. Handling toxins must be avoided since toxins may penetrate through skin or mucous membranes. Prompt washing of the affected area and medical attention are needed for exposure.
Portuguese Man O' War (Physalia physalis)
Portuguese man o' war is a siphonophore of warm sea waters that resembles a jellyfish with its blue-purple, transparent float and trailing, long tentacles. It drifts on ocean surfaces and comes ashore.
Its tentacles bear nematocysts that deliver venom, causing severe pain, welts, and, in some cases, systemic effects like difficult breathing or cardiac irregularities. The venom, a mixture of proteins and peptides, can remain active even after the animal has passed away. Contact requires an immediate rinse with vinegar or sea water (not fresh water) to de-activate stingers, followed by medical treatment for serious reactions.
Lionfish (Pterois sp.)
Lionfish, native to Indo-Pacific oceans but non-native in the Caribbean and Atlantic, inhabit coral reefs and rocky crevices. They have striking red, white, and black stripes and long, fan-shaped pectoral fins and venomous spines.
The spines inject venom which is made up of neurotoxins and acetylcholine, causing extreme pain, inflammation, and, in rare cases, general symptoms like nausea or respiratory impairment. The venom remains potent even after the fish's death. Immersing the affected area in hot (but not boiling) water immediately will denature the venom, and the victim should be treated by a physician in bad instances.
Fat Tail Scorpion (Androctonus australis)
The fat tail scorpion, indigenous to the Middle East and North Africa, lives in scrublands and dry deserts. It has a robust dark brown or black body with a dense tail and tiny pincers, which testifies to its reliance on venom, not brute force.
Its venom, a combination of neurotoxins, targets the nervous system, leading to severe pain, swelling, muscle contractions, and, in extreme instances, respiratory distress or death. The sting is dangerous to children or an allergic person. Prompt medical care is essential, and washing the affected area with soap and water can prevent secondary infections while seeking assistance. brown pods, and have black dots at the top of them. Only one seed, if completely chewed can be fatal to adults and children.
Past Plants and Animals
Plants and animals that have been included in past years' rules are listed below.
2017-2018 Season
Poison Oak (Toxicodendron diversilobum) Poison oak, found throughout western North America in states like California and Washington, is also known as the Pacific poison oak or western poison oak. It can be found in a variety of habitats, ranging from grasslands to conifer forests. It can survive in shady to full sun conditions and prefers elevations below 5000 ft (1500 m). Its form can vary from a dense bush to a vine. During the winter, the stems are leafless; in February, the leaves begin to appear, changing colors from bronze to green to pink. The leaves most commonly have 3 lobes, are 1-4 inches long, and have lobed edges, resembling a very glossy oak leaf. Flowers are white, forming from March to June, and the peak flowering season is in May. Many deer and squirrels consume poison oak with no reactions to the toxin. The leaves and twigs contain urushiol, which causes itching and dermatitis. Burning this plant is very dangerous, as smoke can lead to internal injuries, along with external damage. Death-Cap Mushroom (Amanita phalloides) The death cap mushroom has a large fruiting body, typically 2-6 inches across, that is usually pale green or yellow. It has a distinctive swollen and ragged base (volva) which can be hidden by leaf debris. The scent is often described as overpoweringly sweet. It is widespread across Europe, often found with oaks, chestnuts, and pines. In the US, it can be found on both the East and West Coast. The death cap mushroom, one of the most dangerous mushrooms, is responsible for a large number of mushroom-related deaths each year. This is due to several factors; the death cap mushroom resembles edible fungi like the straw mushroom, and the toxins it contains (called amatoxins) cannot be reduced through cooking. Amatoxins cause renal and hepatic failure (kidney and liver), with symptoms including gastrointestinal distress, jaundice, and cardiac arrest. Treatment includes activated carbon to clear the GI tract and may require a liver transplant if liver failure occurs. Jimson Weed (Datura sp.) Jimson weed, believed to be native to Mexico, is a bush-forming herb with a bad smell. It has a long, thick root, and a branched yellow-green stem. The leaves are long and smooth, with the top side darker than the bottom. The flowers, occurring throughout the summer, are white to purple with a trumpet shape. The flowers generally open at night and have a pleasant smell. It is found throughout most moderate and warm climates, especially near wastelands and roadsides. Its name comes from Jamestown, Virginia, where it was consumed by soldiers during Bacon's rebellion. All parts of this plant contain anticholinergics (atropine, hyoscyamine, scopolamine), causing symptoms such as delirium and tachycardia. Traditionally, it was used for asthma and analgesia, as well as for hallucinogenic effects. Mayapple (Podophyllum peltatum) Mayapple is an herbaceous perennial that can be found throughout the eastern US and southeastern Canada. It grows in colonies that originate from a single root. The stems are generally 30-40 cm tall, with umbrella-like leaves with shallow lobes. The flowers can be white, yellow, or red, appearing in early May. The unripe green fruit is toxic, but the ripe yellow fruit with seeds removed can be safely ingested. However, even large amounts of the ripe fruit can still cause damage. The roots and leaves are poisonous as well. The toxin it contains is known as podophyllotoxin, which can be used topically to treat warts. In traditional medicine, Native Americans used mayapple as an antiemetic and anthelmintic. Ongaonga (Urtica ferox) Ongaonga, native to New Zealand, is a large woody shrub that can grow up to 3 meters tall. Stinging hairs up to 6 mm long cover the stems, leaves, and stalks. The leaves are oppositely arranged and have a triangular shape with serrated margins. It is often found in temperate regions on the edges of forests and flowers from November to March. It provides food and protection for the red admiral butterfly (Vanessa gonerilla) and is eaten by animals such as goats and deer. Ongaonga contains the toxin known as triffydin/tryfydin, which contains histamine, serotonin, and acetylcholine. Reactions range from inflammation to blurred vision and paralysis. One human death from ongaonga contact has been recorded. Cane Toad (Rhinella marina) Cane toads are native to South and Central America. They are night foragers and mainly prey on insects and snails. They have been introduced to various places such as Australia to control insect populations, but for the most part, become an invasive species. Adult cane toads have toxins on glands on their upper surface, especially near the shoulders, and exude bufotoxin, which acts on the heart and central nervous system (CNS), when provoked. Pacific Newt (Taricha sp.) There are four species of Pacific newt: T. granulosa (rough-skinned newt), T. rivularis (red-skinned newt), T. sierrae (Sierra newt), and T. torosa (California newt). All four are found on the Pacific coast, from southern Alaska to southern California. These newts generally have a brown upper body with a brightly colored belly, with granulated or grainy skin. Adults are nocturnal and semi to fully aquatic, while efts (juveniles) are mainly terrestrial. Adults' diets generally consist of invertebrates. All species possess tetrodotoxin, with the rough-skinned newt being the most toxic. However, toxicity can vary, even between the same species living in different regions. Toxins should not come in contact with broken skin or mucous membranes, and hands should be washed after handling to prevent ingestion. Brown Recluse Spider (Loxosceles recluse) The brown recluse is one of three North American spiders with venom that requires medical attention. They are around 6-20 mm long and generally light to dark brown colored. These spiders generally have a marking shaped like a violin on their back, with the neck of the violin pointing towards the spider's rear. Unlike other spiders, which have 8 eyes, brown recluses have 6 eyes that are arranged in pairs. They have a coating of fine hairs that create a soft, furry appearance. Their life span lasts 1 to 5 years, and juveniles take about 1 year to reach maturity. Females lay eggs from May - July in sacs of 50, which hatch in one month. Brown recluses build irregular webs that are located in dry, undisturbed places like garages and cellars. Their diet consists of cockroaches, crickets, and other insects. In North America, brown recluses can be found south of a line roughly connecting southeastern Nebraska to Ohio. Brown recluse bites are often not initially felt, but require medical attention, as their venom is hemotoxic. Symptoms of a bite include nausea, fever, muscle and joint pain, and rashes. The venom can also have necrotic effects, with soft tissue destruction resulting in lasting scars. A typical treatment includes applying an icepack; medications have been used but the bite can usually heal without much intervention. Fattail Scorpion (Androctonus australis) The fattail scorpion is native to North Africa, Somalia, the Middle East, Pakistan, and India. Its exoskeleton is covered in granules, which is hypothesized to allow it to withstand extremely strong sandstorms without digging a burrow. It grows up to 10 cm long with a thick tail and stripes. Although it is one of the most common scorpions in the pet trade, its venom is very potent and causes a number of deaths each year.
|
Common Household Toxins
The 2026 rules of Potions and Poisons mention the following toxic household chemicals:
- Ammonia
- Hydrogen peroxide
- Rubbing alcohol
- Bleach
- Epsom salts
- Vinegar
- Nutritional supplements containing calcium and iron
Ammonia
The compound ammonia itself is a colorless gas with the formula NH3. However, it is most commonly seen in households as a cleaner, where the gas is dissolved into water. It is most dangerous when mixed with bleach, which causes the release of toxic fumes, which can cause serious respiratory damage, in addition to potential chemical burns, headaches, nausea, or vomiting.
Hydrogen peroxide

Hydrogen peroxide is a colorless liquid. Its compound name is H2O2. It is commonly used as a disinfectant, either for surfaces or for wounds. Because the concentration of most hydrogen peroxide used in households (usually in brown bottles) is low, at about 3%, ingestion of small amounts of hydrogen peroxide (diluted) does not usually cause any significant damage, apart from potential stomach irritation. However, ingesting a large quantity can cause more serious stomach irritation and may even cause chemical burns.
Furthermore, ingestion of a higher concentration of hydrogen peroxide can cause much more serious symptoms and death in some cases.
Rubbing Alcohol
Rubbing alcohol (Isopropyl alcohol) is an alcohol with the formula C3H8O. It is most often used as a disinfectant. When ingested, it is metabolized into acetone. This can cause dizziness, headaches, vomiting, or even coma.
Bleach
Bleach is a solution of the chemical compound sodium hypochlorite (NaClO) in water. It is a strong base, with a pH of 12.6. It is most often used as a household cleaner. As mentioned above, mixing bleach with ammonia releases dangerous fumes. Exposure to bleach on its own can cause irritation in the eyes, mouth, skin, and lungs, and can cause burns.
Epsom Salts
Epsom salts (Magnesium sulfate) are salts with the equation MgSO4. They have many uses, including uses as bath salts, as laxatives, face cleansers, cleaners, and as fertilizer. However, ingesting high levels of these salts can cause magnesium overdose, which can lead to slowed heartbeat, lowered blood pressure, nausea, vomiting, and coma or death in serious cases.
Vinegar
Vinegar is an extremely common household chemical. It is an acidic liquid, a mixture of acetic acid (CH3COOH) and water. It is used in cooking, cleaning, and medicine. However, concentrations of acetic acid higher than 10% can cause skin damage/corrosion.
Nutritional Supplements Containing Calcium and Iron
An excess of calcium, known as hypercalcemia, may result from overuse of calcium supplements. Symptoms of hypercalcemia include nausea, thirst, lethargy, and muscle weakness; in severe cases, cardiac arrhythmias and palpitations may occur.
Iron supplements are often taken for anemia, but iron poisoning can occur and be potentially fatal, especially in children under 5 years old. The GI tract and stomach become irritated and internal cell reactions may be interrupted due to an excess of iron. Vomiting and nausea are some of the earliest symptoms; if untreated, the liver may develop severe scars and fail. It can be treated through bowel irrigation or chelation therapy.
Additionally, it is important to note that taking calcium and iron supplements together is not recommended since the calcium will interfere with iron absorption.
Environmental Toxins
This section is incomplete. |
Environmental toxins included in the 2026 season rules include: copper, lead, and mercury.
Copper (Cu)
Copper poisoning usually occurs as a result of drinking contaminated water, copper salt containing creams for treating burns, or cooking acidic food in copper cookware. Symptoms vary based on the method by which one acquires copper toxicity, but the most common method is ingestion, which can lead to abdominal pain, jaundice, thirst, diarrhea, nausea and vomiting. Copper poisoning can also damage the liver and cause the formation of structures in the gastrointestinal tract. Copper salts are also highly toxic to aquatic life.
Lead (Pb)
Lead, a heavy metal, is a highly toxic substance that can cause serious health problems. Exposure to lead can occur through ingestion, inhalation, or skin absorption. Long-term exposure to lead can lead to various health effects, including damage to the nervous system, kidneys, and reproductive system. In children, lead exposure can cause learning disabilities, behavioral problems, and developmental delays. Adults exposed to lead may experience headaches, fatigue, muscle weakness, and abdominal pain. In severe cases, lead poisoning can lead to seizures, coma, and even death. Common sources of lead exposure include contaminated water, lead-based paint, and certain occupational exposures
Mercury (Hg)
Mercury poisoning typically occurs as a result of eating fish, dental fillings, or exposure as a result of work. The symptoms vary based on the amount of mercury a person was exposed to, how long they were exposed to it, and the type of mercury they were exposed to. Typical symptoms include weakness, numbness, anxiety, rashes and skin discoloration, poor vision or hearing, and poor coordination. Kidney problems can also emerge as a long term side effect. Tuna is typically of the greatest concern when it comes to mercury concentration, and consumption should be monitored in young children and nursing mothers. The time it takes for symptoms to appear can vary, taking anywhere from weeks to months for symptoms to appear. Once symptoms appear they increase in severity very rapidly, resulting in a coma or even death.
Past Environmental Toxins
Environmental toxins that have been included in past years' rules are listed below.
2018-2019 Season
Environmental toxins included in the 2019 season rules include: ferric iron, copper, and mercury. Ferric Iron (Fe 3+) Iron poisoning most often occurs when one consumes a large number of iron supplements or pills. Symptoms of iron poisoning are observable around 6 hours after consumption. These symptoms include vomiting, diarrhea, abdominal pain, and dehydration. The main effect of iron poisoning is the corrosion of the intestinal lining. If untreated, it is fatal. Copper (Cu) Copper poisoning usually occurs as a result of drinking contaminated water, copper salt containing creams for treating burns, or cooking acidic food in copper cookware. Symptoms vary based of the method by which one acquires copper toxicity, but the most common method is ingestion, which can lead to abdominal pain, jaundice, thirst, diarrhea, nausea and vomiting. Copper poisoning can also damage the liver and cause the formation of structures in the gastrointestinal tract. Copper salts are also highly toxic to aquatic life.
|
Toxic Spills
Toxic and chemical spills can seriously damage the environment if left unchecked. Not only do the toxins damage the environment they were originally spilled in, they can spread through natural means and damage other environments. The properties of the chemical spilled also have an effect on how it spreads. Chemicals with a higher viscosity or surface tension will be more resistant to movement, and as a result will not spread as easily. These properties also affect how readily an oil will disperse and break down, which can influence local wildlife and how the spill is cleaned up.
Oil may also permeate the environment further through natural processes such as weathering and emulsification. Weathering causes oil to break down and become heavier than water, causing it to be dispersed through a water column or stick to the surface of the water. Evaporation and oxidation also leave behind residues, though with more volatile substances such as gasoline evaporation can remove a significant portion of the oil from the environment. Emulsification results in a mixture of oil and water, lingering in the environment for an extended period of time. This makes the spill much more difficult to clean and can effect the environment for long after the spill takes place.
Regardless of the chemical spilled, organisms in the environment will likely be harmed to some extent after a toxic spill. Food resources and habitats are often destroyed as a result of spills, damaging the ecosystem and its food chain. Food chain issues can affect humans as well, altering where food may be sourced. Physiological problems may also arise as a result of contamination, with some animals suffering damage to the nervous system, liver, or lungs. Reproductive problems may also occur as a result of toxins released into the environment.
Further Reading
Plant pigments: https://docs.google.com/document/d/1tB1gSlES3qCCV_WoruSJgO-Fi_SCXYiljwW1cEHsBb4/edit
SciolyMaster's Notes (Warning: these are from 2019 and therefore may not have all the content in the 2024-2025 rules; despite this, it is a superb resource): https://scioly.org/tests/files/potionspoisons_2019_b_ssss-sciolymaster_notes.pdf
| Division B: Hovercraft · Geocaching · Potions and Poisons | Division C: Code Busters · Mystery Design · Remote Sensing |