Mission Possible/Chemical Devices
Appearance
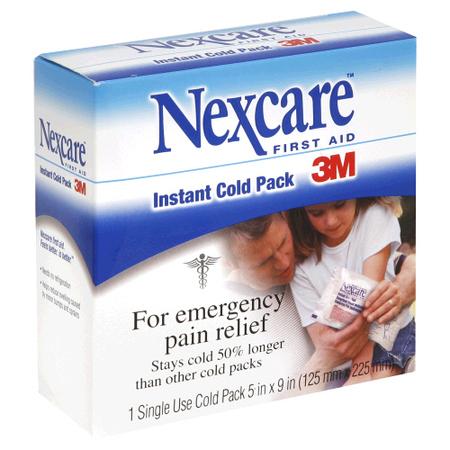
Endothermic Reactions
Ammonium Chloride and Water
Exothermic Reactions
Oxidizing Iron
 |
These one-use packets oxidize iron filings to produce heat.. |
Gas Producing Reactions
Vinegar and Baking Soda
 |
The classic elementary science project volcano components. Mix vinegar and baking soda to generate carbon dioxide gas. |
Alka Seltzer and Water
Mentos and Coke
| Potentially much more dramatic than other gas producing reactions. Either use small quantities, contain it well, or both. If anything leaves the boundaries of the machine, you will be penalized. |